|
9/6/2023 0 Comments Dipole moment of co2 The order of decreasing dipole moment is CH3Cl > CH2Cl2 > CHCl3 > CCl4. How do you find the dipole moment of ch3cl? What is dipole moment in chemistry class 11?ĭipole moment is the product of electric charge and distance between the positive and negative species present in the molecule. The calculated or observed dipole moment = 1.03 Debye = 1.03 × 10−18 esu cm. If we consider HCl as a purely ionic compound the charge on hydrogen and chlorine = 4.8 × 10−10 esu and bond length = 1.27 × 10−8 cm. How do you find the dipole moment of HCl? The net dipole moment of three N- H bonds will add up to give 1.4D. In NH3, we know nitrogen is more electronegative than hydrogen, so the dipole moment is from H to N. How do you find the dipole moment of nh3? Therefore the net dipole moment of the water molecule = 0.9187+0.9187=1.837D. – We have to calculate the individual dipole moments and later we have to do the sum to get the dipole moment of the water molecule. – There are two hydrogen in water molecules, the two hydrogen creates their individual dipole moments. How do you find the dipole moment of H2O? Thus, e (charge) can be determined in esu. From the bond angle and vector moment mu = e × l. If bond angle is 1.5^o and O – H bond length is 0.94 A, determine magnitude of the charge on the oxygen atom in the water molecule. What is the dipole moment of H2O?ĭipole moment of H2O is 1.85 D. So, CO2 has zero dipole moment because it is a linear molecule. But, as the dipole moment of one bond is cancelled by other, the structure of CO2 is a linear. in which there is bond dipole moment (C-O bond). How do you calculate the dipole moment of CO2?ĬO2 has the structure as O=C=O. The bond dipole moment is a vector quantity therefore it has both magnitude and direction both. It is a measure of the separation of negative and positive charges in a system. Bond dipole involves the concept of electric dipole moment. 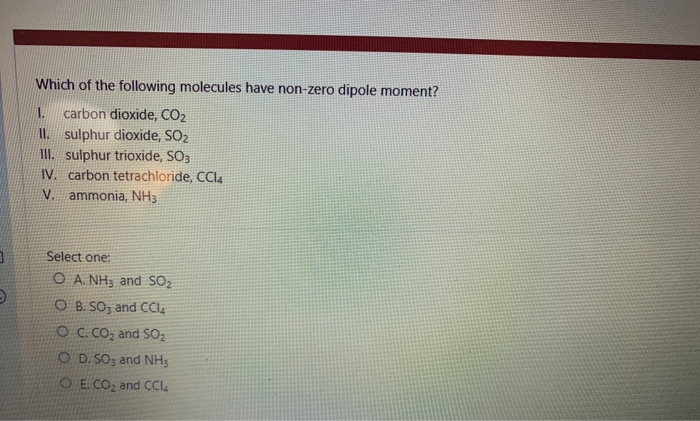
What is the dipole moment in chemistry?Ī bond dipole moment is a measure of the polarity between two atoms in a molecule. 
By convention the vector a points from the negative to the positive charge. The electric dipole moment is defined as p = qa. 24 Why is the dipole moment of bf3 zero? What is dipole formula?Īn electric dipole is formed by two point charges +q and −q connected by a vector a. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
